Abstract #: 1376
Authors: Haley E. Ramsey1-2, Maria P. Arrate1, Londa Fuller1, Agnieszka E. Gorska1, Kelli Boyd3-4, Melissa A. Fischer1-2, Aram Oganesian5, Mohammad Azab5, and Michael R. Savona1,2,4
1Department of Medicine, 2Cancer Biology Program, 3Department of Pathology, Microbiology and Immunology, 4Vanderbilt-Ingram Cancer Center; Vanderbilt University School of Medicine, Nashville, TN USA; 5Astex Pharmaceuticals, Inc., Pleasanton, CA
Background: DNA methyltransferase inhibitors (DNMTi) induce remissions and improve survival for patients with MDS and those with AML unable to receive standard cytotoxic chemotherapy. Accordingly, DNMTi therapy is the backbone of SOC treatment for MDS and AML. Given the inconvenience, pain, and general detriments to QOL with SQ or IV therapy daily for 5-7 days every month with azacitidine (AZA) or decitabine (DAC), many have attempted to provide the therapy orally, but encountered difficulties with this method of administration given rapid first-pass clearance via the enzyme cytidine deaminase (CDA) which is ubiquitous in the gut and liver. Recently, DAC was combined with cedazuridine (CDZ), an oral CDA inhibitor, in a fixed-dose (35 mg/100 mg) combination tablet (ASTX727) to approximate the pharmacokinetics of IV DAC (Savona et al Lancet Haematology 2019). To determine if a similar strategy might be feasible with AZA, we attempted to increase the bioavailability of oral AZA with CDZ in a murine tumor model.
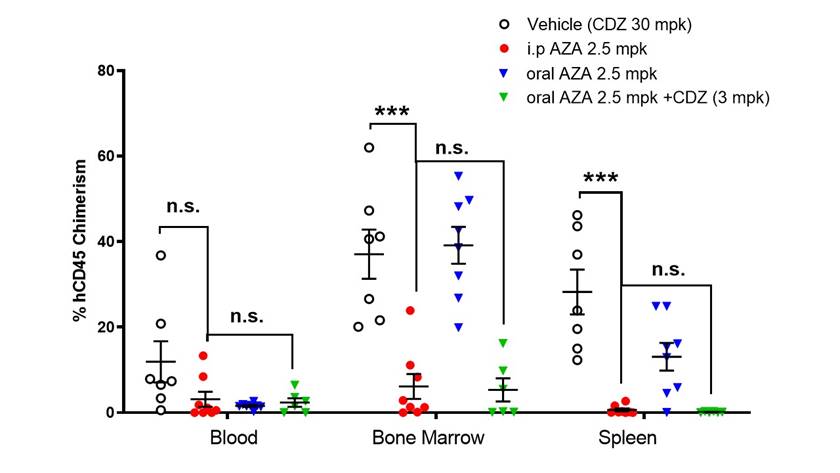
Methods: We measured GI50 in AML cell lines treated with vehicle (DMSO), AZA, and AZA/CDZ combination. Although cancer cell lines produce CDA, total levels are negligible in comparison CDA in the gut and liver. For this reason, we then studied CDZ, AZA, and the AZA/CDZ combination in a systemic model of AML in immunocompromised mice. NSGS mice were sub-lethally irradiated and administered MOLM-13 AML cells via tail vein injection. At day seven post-transplant, engrafted mice were randomized to receive 2.5 mpk i.p. AZA(n=8), 2.5 mpk oral AZA(n=8), 3 mpk CDZ + oral AZA(n=6), or CDZ alone at 30 mpk (n=7). All oral AZA and CDZ was administered via oral gavage daily for 7 consecutive days; i.p. therapy was similarly given for 7 consecutive days. During treatment, the kinetics of MOLM-13 expansion was defined by detection of human AML in the blood as detected by flow cytometry. At approximately three weeks after transplant, CDZ-only treated mice became moribund, and all experimental groups were sacrificed for analysis of chimerism.
Results: After 72 hour of treatment, no differences were noted in viability of cell lines between AZA and AZA+ CDZ in vitro. In the xenograft model, as expected, i.p. AZA-treated mice had significant decreases in leukemic expansion in the bone marrow and spleen, whereas CDZ alone did not (AZA i.p. vs CDZ, p = 0.004 and <0.001). More importantly, whereas oral AZA alone failed to decrease AML expansion in both the bone marrow and spleen of treated mice (oral AZA vs CDZ., p = 0.677 and 0.249, respectively), the addition of CDZ to oral AZA led to significant decreases in AML in both bone marrow and spleen (Oral AZA + CDZ vs CDZ, p = 0.012 and 0.004, respectively). Likewise, addition of CDZ to oral AZA showed no significant differences in activity against AML compared to traditional AZA i.p. dosing in either bone marrow or splenic tissue (p= 0.204 and 0.224, respectively). Furthermore, in a Kaplan-Meier survival analysis, mice treated with CDZ alone die within 21 days of transplant, but both i.p. AZA and oral AZA + CDZ treated mice had a 50% extended survival with no significant difference in survival between i.p. AZA and oral AZA + CDZ treated mice (p= 0.502). H&E staining revealed no significant bone marrow toxicity in treated mice, suggesting that AZA preferentially effected transplanted MOLM-13 AML cells. Conclusion: Consistent with previous preclinical and clinical studies with ASTX727, the oral AZA approximated AZA anti-tumor activity when co-treated with CDA-inhibitor CDZ. These preliminary data provide rationale for the development of CDZ + oral AZA therapy as a fixed dose combination (ASTX030) in myeloid disease. Clinical trials evaluating ASTX030 are being planned.